Treating PFAS in Sludge
Rising concerns over Poly- and Perfluorinated Alkylated Substances (PFAS) contamination of water and soils have sparked interest in this emerging class of contaminants as it pertains to wastewater sewage sludge. Two typesof PFAS have been linked to increasing concern for human health and environmental impacts, namely perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), particularly in regards to their persistent and bioaccumulative nature.
Due to the extensive use of PFAS compounds for over eight decades, including in everyday household items such as food packaging, cookware, stain resistant clothing and industrial applications like electroplating and fire fighting foam, it is generally accepted that nearly all wastewater treatment sludges will have some trace background level of PFAS compounds.
Wastewater sludges have been found to have perfluoroalkyl carboxylates (PFCAs) and perfluoroalkane sulfonates (PFSAs), and additional compounds such as GenX are being detected in liquids and solids as analytical methods improve.
While the concentrations of measurable PFAS compounds in sludge are relatively low, the need to identify a feasible option for the ultimate disposal of PFAS contaminated sludge is warranted.
While the concentrations of measurable PFAS compounds in sludge are relatively low, the need to identify a feasible option for the ultimate disposal of PFAS contaminated sludge is warranted.
Traditionally in the US, sludge is alkaline-stabilized and land applied. The alkaline stabilization process involves the addition of lime to wastewater sludge and raising the pH to ~12, for a contact period no less than 2 hours, with numerous other operational methods to yield varying degrees of treatment. The resulting sludge quality varies, but in general, the technique is only capable of removing fecal coliform and salmonella, but not ova of parasites or more complex compounds such as PFAS.
Alternatively, sludge can be digested aerobically or anaerobically at varying temperature ranges to successfully eliminate ova of parasites, but neither of these processes are known to remove PFAS compounds. These methods pose a potential threat in terms of PFAS exposures as these compounds can exit those processes and transport into the natural environment when biosolids, although stabilized to Class A or EQ quality, are land applied or landfilled.
Current research efforts are indicating that combustion processes at higher temperatures can be adapted to mitigate these issues and properly address the challenge of evolving emission standards.
With thermal oxidation as the leading contender for the treatment of PFAS compounds in sludge, the focus of this research turned to established technologies that can be readily deployed to address the market's needs. Veolia Water Technologies has developed and successfully installed a sustainable drying and combustion system in the municipal wastewater management market using a system called BioCon™ ERS. This system combines the efficiencies of a convective air medium temperature belt dryer with a biomass furnace. The purpose is to completely combust all organic material and recover sensible heat. The heat available in the exhaust is captured for use as process heat in the belt dryer. This integrated dryer and energy recovery system is sustained by heat generated from the combustion of biosolids to drive the drying process, and in turn the dried material fuels the combustion system, making it a fully energy-sufficient and sustainable system.
While the use of BioCon™ ERS for PFAS treatment is novel, the concept of combining these two technologies is not. For example, the City of Buffalo, MN has been beneficially using their biosolids as a fuel source at the Buffalo Wastewater Treatment Facility (WWTF) using this process scheme since 2008. The solids treatment system at Buffalo WWTF consists of both BioCon™ thermal drying and ERS thermal oxidation processes to reduce the amount of material leaving the site while minimizing overall fossil fuel consumption.
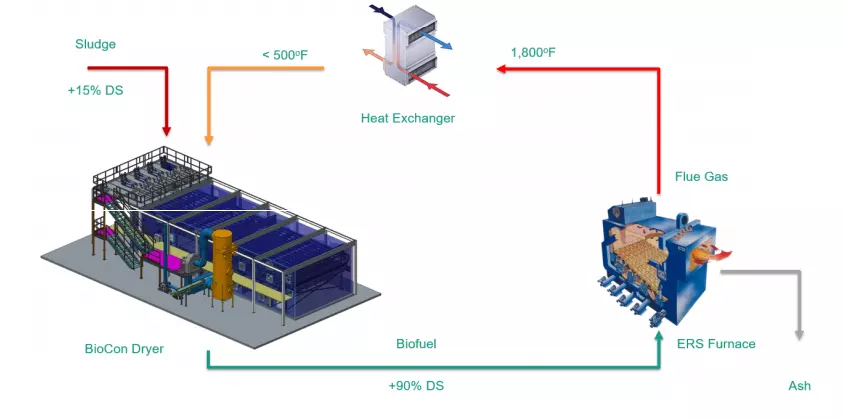
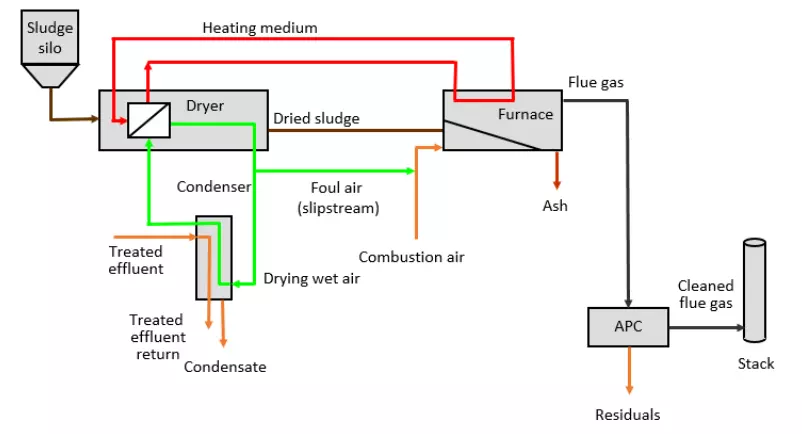
The thermal treatment process consists of several steps as depicted in the process flow diagram. Dewatered solids are pumped from a storage silo, into the dryer cabinet through oscillating depositors, and onto a slowly moving belt inside the dryer. Heat is transferred to the solids by circulating air between an air-to-air heat exchanger that recovers energy from the furnace flue gas and the dryer cabinet. Moisture is removed from the drying air by continuously extracting a portion of the air from the dryer, transferring it through a condenser and returning it back to the dryer.
A portion of the air from the condenser is removed from the system to maintain a negative pressure on the drying system. The foul air is blended with fresh air and used as combustion air in the burners and furnace eliminating the need for separate odor control systems.
Knowing the flexibility this combined system offers, the research turned the focus on determining the efficacy of treating PFAS laden sludge using this system that has been proven for over a decade.
Led by Veolia, a bench scale oxidative study was undertaken in collaboration with the University of Dayton Research Institute in Dayton, Ohio. The goal was to study the fate and transport of PFAS in sludge undergoing full combustion and its impact on solids residue, process liquids and air emissions. The experimental setup included a thermal reactor that is a batch-charged, continuous flow reactor system. This system was configured to represent gas-phase behavior in a full-scale waste incinerator prior to air pollution controls. This set up allows the study of combustion at various conditions, including adjustments to temperature, oxygen applied and dwell time to mimic controllable conditions within the ERS.
The study was conducted in two phases. Phase 1 involved the determination of transport efficiency of the laboratory setup to ensure PFAS can be captured for analytical confirmation. Phase 2 involved the thermal oxidation of PFAS laden sludge to determine if specific PFAS can be decomposed at designated temperatures that mimic a full-scale ERS biomass furnace. Results from Phase 1 suggest that transport efficiency is greater than 80%. Results from the Phase 2 study demonstrate that complete degradation of certain PFAS compounds under optimal thermal oxidation is possible.
Based on results obtained and decomposition rates calculated thus far, PFAS decomposition using the proposed treatment train is achievable and exceeds treatment objectives. An attempt to develop a Fluorine mass balance is underway. Due to the highly reactive nature of hydrogen fluoride (HF) and the low LOQ of HF analysis, it is possible that a fluorine mass balance could result in poor outcome or is unachievable. Results from this study are being used to industrialize a full-scale thermal oxidation solution for the treatment of PFAS laden sludge. Veolia continues with studies aimed at fine-tuning the industrial scale treatment scheme and further optimization of the air pollution control scheme are underway.
_______________________
The information contained in this statement is based on the Veolia group's understanding and know-how of the scientific and technical fields discussed herein as of the time of publication. Statements that may be interpreted as predictive of future outcomes or performance should not be considered guarantees of such, but rather reasoned assessments of the possible evolution of the technologies described.
As this document is based on the state of the Veolia group's scientific, technical, and regulatory knowledge at the time of its publication, the completeness and accuracy of the information contained herein cannot be guaranteed.
Descriptions contained herein apply exclusively to those examples and/or to the general situations specifically referenced, and in no event should be considered to apply to specific scenarios without prior review and validation.


